3 results
Integrated efficacy analysis from phase 3 studies of investigational microbiome therapeutic, SER-109, in recurrent Clostridioides difficile infection
- Matthew Sims, Michael Silverman, Thomas Louie, Elaine Wang, Colleen Kraft, Mayur Ramesh, Tatiana Bogdanovich, Kelly Brady, David Lombardi, Asli Memisoglu, Ananya De, Brooke Hasson, Christine Lee, Paul Feuerstadt, Darrell Pardi, Colleen Kelly, Peter Daley, Godson Oguchi, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
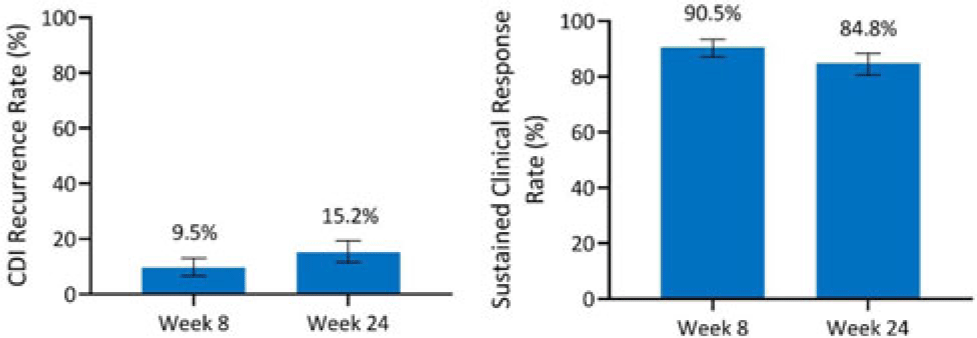
Background: Antibiotics alone are often insufficient to treat recurrent C. difficile infection (rCDI) because they have no activity against C. difficile spores that germinate within a disrupted microbiome. SER-109, an investigational, oral, microbiome therapeutic comprised of purified Firmicutes spores, was designed to reduce rCDI through microbiome repair. We report an integrated efficacy analysis through week 24 for SER-109 from phase 3 studies, ECOSPOR III and ECOSPOR IV. Methods: ECOSPOR III was a randomized, placebo-controlled phase 3 trial conducted at 56 US or Canadian sites that included 182 participants with ≥2 CDI recurrences, confirmed via toxin EIA testing. Participants were stratified by age (<65 years or ≥65 years) and antibiotic regimen (vancomycin, fidaxomicin) and were randomized 1:1 to placebo or SER-109 groups. ECOSPOR IV was an open-label, single-arm study conducted at 72 US or Canadian sites including 263 participants with rCDI enrolled in 2 cohorts: (1) rollover participants from ECOSPOR III who experienced on-study recurrence diagnosed by toxin EIA (n = 29) and (2) participants with ≥1 CDI recurrence (diagnosed by PCR or toxin EIA), inclusive of the current episode (n = 234). In both studies, the investigational product was administered orally as 4 capsules over 3 consecutive days following symptom resolution after standard-of-care antibiotics. The primary efficacy end point was rCDI (recurrent toxin-positive diarrhea requiring treatment) through week 8. Other end points included CDI recurrence rates and safety through 24 weeks. Results: These 349 participants received at least 1 dose of SER-109 in ECOSPOR III or ECOSPOR IV (mean age 64.2; 68.8% female). Overall, 77 participants (22.1%) enrolled with their first CDI recurrence. Four participants received blinded SER-109 in ECOSPOR III followed by a second dose of open-label SER-109 in ECOSPOR IV. Overall, the proportion of participants who received any dose of SER-109 with rCDI at week 8 was 9.5% (33 of 349; 95% CI, 6.6 %–13.0%), and the CDI recurrence rate remained low through 24 weeks (15.2%, 53 of 349; 95% CI, 11.6%–19.4%), corresponding to sustained clinical response rates of 90.5% (95% CI, 87.0%–93.4%) and 84.8% (95% CI, 80.6%–88.4%), respectively (Fig. 1). Most rollover participants (25 of 29, 86.2%) were from the placebo arm; 13.8% had rCDI by week 8. Conclusions: In this integrated analysis, the rates of rCDI were low and durable in participants who received the investigational microbiome therapeutic SER-109, with sustained clinical response rates of 90.5% and 84.8% at weeks 8 and 24, respectively. These data further support the potential benefit of microbiome repair with SER-109 following antibiotics for rCDI to prevent recurrence in high-risk patients.
Financial support: This study was funded by Seres Therapeutics.
Disclosure: None
Integrated safety analysis of phase 3 studies for investigational microbiome therapeutic, SER-109, in recurrent CDI
- Matthew Sims, Charles Berenson, Stuart Cohen, Elaine Wang, Elizabeth Hohmann, Richard Nathan, Alberto Odio, Paul Cook, Kelly Brady, David Lombardi, Asli Memisoglu, Ananya De, Brooke Hasson, Bret Lashner, Louis Korman, Doria Grimard, Juan Carlos Moises Gutierrez, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s44-s45
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Clostridioides difficile infection (CDI) often recurs in patients aged ≥65 years and those with comorbidities. Clinical trials often exclude patients with history of immunosuppression, malignancy, renal insufficiency, or other comorbidities. In a phase 3 trial (ECOSPOR III), SER-109 was superior to placebo in reducing recurrent CDI (rCDI) risk at week 8 and was well tolerated. We report integrated safety data for SER-109 in a broad patient population through week 24 from phase 3 studies: ECOSPOR III and ECOSPOR IV. Methods: ECOSPOR III was a double-blind, placebo-controlled trial conducted in participants with ≥2 CDI recurrences randomized 1:1 to placebo or SER-109. ECOSPOR IV was an open-label, single-arm study conducted in 263 patients with rCDI enrolled in 2 cohorts: (1) rollover participants from ECOSPOR III with on-study recurrence and (2) participants with ≥1 CDI recurrence, inclusive of the current episode. In both studies, the investigational product was administered as 4 oral capsules over 3 days. Treatment-emergent adverse events (TEAEs) were collected through week 8; serious TEAEs and TEAEs of special interest (ie, bacteremia, abscess, meningitis) were collected through week 24. Results: In total, 349 participants received SER-109 in ECOSPOR III and/or ECOSPOR IV (mean age 64.2; 68.8% female). Chronic diseases included cardiac disease (31.2%), immunocompromised or immunosuppressed (21.2%), diabetes (18.9% ), and renal impairment or failure (13.2%). Overall, 221 (63.3%) of 349 participants who received SER-109 experienced TEAEs through week 24. Most were mild to moderate and gastrointestinal. The most common (>5% of participants) treatment related TEAEs were flatulence, abdominal pain and distension, decreased appetite, constipation, nausea, fatigue, and diarrhea. No participants experienced a treatment-related TEAE leading to study withdrawal. Invasive infections were observed in 28 participants (8%); those with identified pathogens were unrelated to SER-109 species, and all were deemed unrelated to treatment by the investigators. There were 11 deaths (3.2%) and 48 participants (13.8%) with serious TEAEs, none of which were deemed treatment related. There were no clinically important differences in the safety profile across subgroups of sex, race, prior antibiotic regimen, or number of CDI recurrences. No safety signals were observed in participants with renal impairment or failure, diabetes, cardiac disease, or immunocompromised or immunosuppressed individuals. Conclusions: In this integrated analysis of phase 3 trials, SER-109, an investigational microbiome therapeutic, was well tolerated in this vulnerable patient population with prevalent comorbidities. No infections, nor those with identified pathogens, were attributed to SER-109 or product species. This safety profile might be expected because this purified product is composed of spore-forming Firmicutes normally abundant in the healthy microbiome.
Financial support: This study was funded by Seres Therapeutics.
Disclosures: None
50 Adjunctive Buprenorphine/Samidorphan Combination in Patients with Major Depressive Disorder: Phase 3 Long-term Extension Study Results
- Michael Thase, Arielle D. Stanford, Asli Memisoglu, William Martin, Amy Claxton, J. Alexander Bodnik, Madhukar H. Trivedi, Maurizio Fava, Sanjeev Pathak
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 12 March 2019, pp. 203-204
-
- Article
-
- You have access Access
- Export citation
-
Introduction
Buprenorphine/samidorphan (BUP/SAM), a combination of BUP (a µ-opioid receptor partial agonist and κ-antagonist) and SAM (a sublingually bioavailable µ-opioid antagonist), is an investigational opioid system modulator for depression. BUP/SAM has shown efficacy versus placebo as an adjunctive treatment for major depressive disorder (MDD) and a consistent safety profile in previously reported, placebo-controlled clinical studies.1,2
Study Objective(s)1. To characterize the safety profile following long-term treatment with BUP/SAM
2. To explore depression symptoms and remission rates in patients with MDD following long-term treatment with BUP/SAM
MethodsFORWARD-2 (Clinicaltrials.gov ID: NCT02141399) enrolled patients who had participated in 1 of 4 controlled studies as well as de novo patients. All patients had a confirmed diagnosis of MDD, had a history of inadequate response to standard antidepressant therapies (ADTs), and had been treated with an adequate dose of an established ADT for ≥8weeks before BUP/SAM initiation. ADT dosage could be titrated, but the ADT could not be changed. During the study, patients received open-label, sublingual BUP/SAM 2mg/2mg as adjunctive treatment for up to 52weeks. Safety (primary objective) was assessed via adverse events (AEs), vital signs, laboratory analytes, and electrocardiography. Suicidal ideation or behavior (SIB) was evaluated by the Columbia Suicide Severity Rating Scale. Abuse potential, dependence, and withdrawal were assessed by AEs and the Clinical Opiate Withdrawal Scale. Exploratory efficacy endpoints included mean Montgomery–Åsberg Depression Rating Scale (MADRS) scores and remission rate (MADRS ≤10).
ResultsOf 1454 total patients, 49% completed the 52-week study, 11% discontinued due to an AE, and 40% discontinued because of other reasons as of the interim data cutoff date (April 30, 2017). Most AEs were of mild/moderate severity. Serious AEs were reported in 3.2% of patients. AEs occurring in ≥10% of patients were nausea, headache, constipation, dizziness, and somnolence. There was no evidence of increased risk of SIB with BUP/SAM. Incidence of euphoria-related events was low (1.2%). After abrupt discontinuation of BUP/SAM, there was little evidence of withdrawal. BUP/SAM was not associated with meaningful changes in laboratory or metabolic parameters or in bodyweight. The mean MADRS score decreased from 22.9 (±9.7) at baseline to 9.8 (±8.8) after 52weeks. The remission rate at 52weeks was 52.5%.
ConclusionsLong-term treatment with BUP/SAM did not reveal any new safety findings and confirmed that the risk of abuse and dependence with BUP/SAM was low. BUP/SAM maintained an antidepressant effect for up to 52weeks of treatment in patients with MDD.
Funding Acknowledgements: Alkermes, Inc.




